



CLABSI Prevention Device
Senior CapstoneA handheld UV-C disinfection device for central-line hubs, developed at Duke University's Pratt School of Engineering.
View on GitHubProblem & Need
Central Line-Associated Bloodstream Infections (CLABSIs) occur when bacteria enter the bloodstream via a central line. 40,000 CLABSIs occur annually in the US, costing hospitals approximately $1.9B per year — and 65–70% are completely preventable with proper disinfection.
The current standard of care is the Scrub-the-Hub technique, which relies entirely on nurse compliance with manual protocol. Infections arise from human error and inconsistent adherence to workflow. This device removes that dependency.
Need Statement: Hospital nurses need a quick, reliable, and user-friendly system to disinfect central lines to maintain adherence to proper protocols and reduce infection risk in patients.
Why UV-C Disinfection?
DNA and RNA strongly absorb 260 nm UV-C incident light. The high-energy photons create nitrogenous base lesions, distorting the helix structure and blocking transcription and translation — effectively sterilizing the hub surface without chemicals, contact, or manual technique.
Device Design
Mechanical
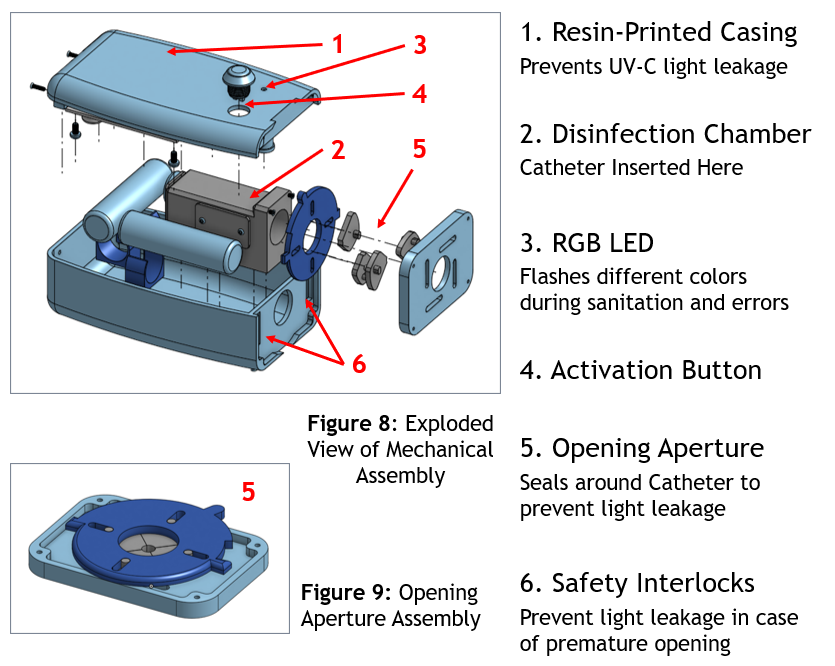
The enclosure is resin-printed to prevent UV-C light leakage. Key mechanical elements:
| # | Component | Role |
|---|---|---|
| 1 | Resin-Printed Casing | Contains UV-C, prevents leakage |
| 2 | Disinfection Chamber | Where the catheter hub is inserted |
| 3 | RGB LED | Color-coded status during operation and errors |
| 4 | Activation Button | Single-button start/abort |
| 5 | Opening Aperture | Seals around the catheter to prevent leakage |
| 6 | Safety Interlocks (×2) | Abort if device opens prematurely |
Electrical
Three custom KiCad PCBs make up the electrical system:
| Board | Function |
|---|---|
| Main | nRF54L15 MCU, power management, BMS (MAX17260), RGB LED driver (LP5815) |
| UVC | UV-C LED driver with current-limiting elements |
| Base | Mechanical base and connector board |
Firmware
Written in C on Zephyr RTOS (nRF Connect SDK), targeting the nRF54L15. The application runs a hierarchical state machine (Zephyr SMF):
INIT → IDLE ──► DISINFECTING ──► IDLE
──► CHARGING ──► (abort via button or interlock)
──► ERROR| State | Behavior |
|---|---|
INIT | Initialize GPIO, PWM, I2C, BMS, safety interlocks |
IDLE | Awaiting button press; battery SOC displayed on RGB LED |
CHARGING | Charge in progress; RGB indicates charge level |
DISINFECTING | UV-C LED at full PWM; instant abort on interlock open or button press |
ERROR | RGB error pattern + buzzer; cleared by button press |
The critical safety path: interlock GPIO interrupts submit a work item that disables PWM output before the SMF state transition completes — mean shutoff time of 17 ± 2 ms, well within the 450 ms safety limit derived from ISO 15858.
Testing Results
| Metric | Value |
|---|---|
| Device Weight | 204 g |
| UV-C LED Optical Power | 2.49 ± 0.047 mW |
| UV-C LED Bandwidth | 262.9 – 273.2 nm |
| Rapid Shutoff Time | 17 ± 2 ms (limit: <450 ms) |
| Germicidal CFU Reduction | 4-log reduction (n=3 replicates) |
Germicidal testing used DH5α bacteria incubated, seeded, and adhered to catheter hubs at room temperature. Hubs were irradiated with UV-C at 0, 2, and 4 hours, then vortexed for CFU counts by serial dilution — achieving a 4-log reduction.
Regulatory & Market
The CLAB-Free device will be classified as a Class II FDA device given the inherent risk of UV light and catheter use. Two potential pathways:
- 510(k) — Preferred: If determined similar enough to existing UV surface-disinfecting devices
- De Novo: If disinfection through catheter tubing constitutes a novel use case
Conclusion & Next Steps
The device successfully disinfects central line hubs using UV-C light in the germicidal range, achieves a 4-log CFU reduction, and operates safely within ISO 15858 exposure limits. It is portable, one-handed, and reusable between patients.
Planned next steps:
- Nurse usability testing
- Design of docking/recharging stations
- Autoclavable disinfection chamber redesign
- Head-to-head testing against proper and improper Scrub-the-Hub technique
Project Documents
Final Design History File
Duke BME capstone poster
KiCad interactive bill of materials — click components to highlight on the PCB
Fab-ready Gerber exports for all three PCB boards
Final revision schematic, PCB layout, BOM, and 3D STEP models
Zephyr RTOS application for the nRF54L15 MCU